Written by VERIFIED Credit Card Processing | High-Risk Payments Specialist
Reviewed by the VERIFIED Underwriting & Compliance Risk Team
Most supplement merchant account problems do not begin with fraud. They begin with underwriting risk perception. An FDA warning letter can dramatically change how acquiring banks, payment processors, and underwriting teams evaluate a supplement business — even when the merchant is still operating legally.
That distinction matters.
Payment processors are not FDA regulators. They evaluate supplement merchants primarily through the lens of financial and reputational risk. When a supplement company receives an FDA warning letter, banks often interpret it as a signal that future disputes, compliance issues, reputational exposure, or card network pressure could increase. That can lead to reserve increases, enhanced monitoring, delayed approvals, or full merchant account shutdowns.
For supplement merchants, understanding how processors evaluate FDA-related risk is now part of protecting long-term payment stability.
Last Updated: May 2026
A FDA warning letter supplement merchant account issue occurs when a supplement business receives FDA scrutiny and its payment processor or acquiring bank reassesses the merchant account for underwriting, compliance, chargeback, or reputational risk.
Key Highlights
- FDA warning letters often increase underwriting scrutiny for supplement and nutraceutical merchants.
- Processors evaluate warning letters as future financial risk signals — not simply regulatory events.
- Claims language, weight-loss marketing, recurring billing, and aggressive funnels commonly trigger processor concern.
- Most supplement merchant account shutdowns occur during post-approval monitoring rather than onboarding.
- Visa monitoring programs often intensify once merchants approach excessive chargeback thresholds.1
- Merchants can often regain stable processing after remediation if underwriting is handled correctly.
What Is an FDA Warning Letter?
An FDA warning letter is a formal notice issued by the U.S. Food and Drug Administration indicating that a company may be violating federal regulations related to product claims, labeling, manufacturing, or marketing. In payment processing, warning letters are often interpreted by acquiring banks as elevated underwriting risk because they can predict future disputes, reputational exposure, or card network scrutiny.
Most warning letters in the supplement industry involve:
- disease-treatment claims
- misleading marketing language
- unapproved drug-like positioning
- manufacturing concerns
- adverse event reporting issues
- labeling deficiencies
The key underwriting reality is this:
Banks do not evaluate warning letters like regulators. They evaluate them like future loss predictors.
That changes how merchant accounts are monitored after a warning letter becomes public.
Why Payment Processors Care About FDA Warning Letters
Payment processors care about FDA warning letters because they increase the perceived probability of future financial exposure.
From an underwriting perspective, warning letters can suggest:
- future customer disputes
- refund spikes
- chargeback increases
- consumer complaints
- card network monitoring
- reputational damage
- legal escalation risk
- higher compliance oversight costs
Underwriters think probabilistically. They are not asking whether a merchant is “good” or “bad.” They are asking whether the merchant account is becoming more difficult or expensive to support.
That is why some supplement businesses continue processing after a warning letter while others experience reserves, monitoring programs, or sudden account termination.
For merchants already under processor scrutiny, this breakdown of supplement merchant account shutdowns explains how underwriting escalation typically unfolds.
How FDA Warning Letters Affect Merchant Accounts
Reserve Increases
One of the most common processor reactions is increasing rolling reserves.
If a supplement merchant previously operated with a 5% reserve, the acquiring bank may increase it to 10–15% after compliance concerns emerge. Exact reserve structures vary by sponsor bank, product category, billing model, and historical dispute ratios.
This helps processors offset potential future losses from:
- chargebacks
- refund surges
- regulatory fallout
- network penalties
Enhanced Monitoring
Many processors place merchants into heightened review programs after warning letters appear publicly.
This may include:
- manual transaction reviews
- monthly underwriting updates
- website rescans
- chargeback ratio monitoring
- traffic-source analysis
- marketing audits
Visa’s dispute monitoring frameworks have increased processor sensitivity around sustained dispute activity, particularly for merchants approaching excessive chargeback thresholds.2
Monitoring often intensifies when chargebacks trend toward 0.75%–0.9%, even before formal network thresholds are crossed.
Onboarding Declines
Some processors immediately decline new supplement applications associated with recent FDA warning letters.
Others may still approve the account — but only after:
- claims remediation
- website revisions
- product restrictions
- underwriting clarification
- enhanced reserves
This is why underwriting-aware placement matters so heavily in supplements.
Merchant Account Shutdowns
In more aggressive scenarios, processors terminate the merchant account entirely.
This is especially common when warning letters coincide with:
- high chargeback ratios
- negative publicity
- continuity billing complaints
- affiliate abuse
- aggressive weight-loss marketing
- hidden subscription terms
Most supplement shutdowns are not caused by a single issue.
They are caused by cumulative risk signals.
Common Supplement Claims That Trigger Processor Concern
Weight Loss Claims
Weight-loss supplements consistently attract heightened underwriting scrutiny.
Phrases like:
- “rapid fat burning”
- “clinically proven weight loss”
- “Ozempic alternative”
- “melt stubborn fat fast”
often trigger both regulatory and processor concern.
Underwriters associate aggressive weight-loss positioning with elevated dispute rates and reputational exposure.
Medical or Disease Claims
Statements implying treatment, prevention, or cure of disease are among the fastest ways to trigger reviews.
Examples include:
- diabetes treatment language
- arthritis relief claims
- blood pressure positioning
- depression or anxiety treatment wording
The FDA regularly issues warning letters involving disease-treatment positioning in dietary supplement marketing.3
Before-and-After Marketing
Before-and-after imagery creates major processor sensitivity because it often correlates with elevated customer expectations and higher refund activity.
Processors frequently review:
- landing pages
- affiliate funnels
- social creatives
- email marketing
- influencer campaigns
Continuity Billing Models
Recurring billing dramatically changes underwriting risk.
Subscription-based supplement merchants face closer scrutiny because continuity models historically produce higher dispute ratios.
Underwriters evaluate:
- rebill clarity
- checkout disclosure
- cancellation friction
- descriptor accuracy
- trial language
How Banks Actually Evaluate Supplement Merchants
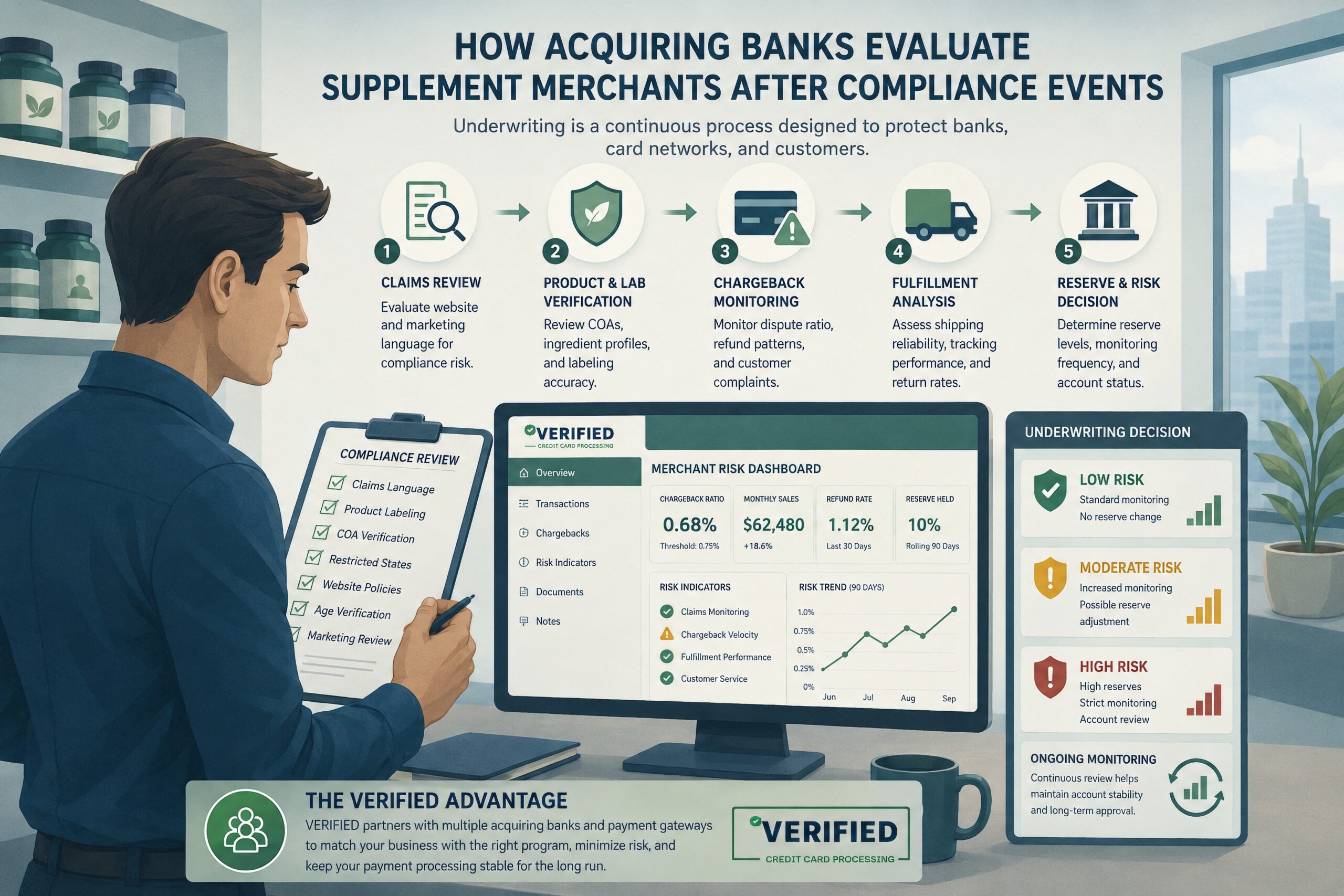
Most merchants think underwriting is about documents.
In reality, supplement underwriting is behavioral analysis.
Processors evaluate whether a merchant account is likely to remain stable over time.
| Underwriting Area | What Banks Evaluate | Why It Matters |
|---|---|---|
| Claims Language | Medical positioning, weight-loss claims, testimonials | Predicts disputes and compliance exposure |
| Chargebacks | Current ratios and trend direction | Indicates future financial liability |
| Traffic Sources | Affiliate quality and ad aggressiveness | Poor traffic often increases refund rates |
| Billing Model | Subscription terms and rebill transparency | Continuity risk affects network exposure |
| Fulfillment | Shipping consistency and support responsiveness | Non-receipt disputes increase processor losses |
| Compliance History | Warning letters or prior shutdowns | Signals elevated monitoring risk |
Underwriters monitor operational behavior more than merchant promises.
That is why some merchants remain stable despite prior compliance issues while others fail quickly after approval.
Operational Example: How Risk Escalates
A supplement merchant may receive an FDA warning letter for aggressive disease-treatment claims while still maintaining stable payment processing initially. If the merchant quickly removes claims language, improves customer support responsiveness, and keeps chargebacks controlled, many acquiring banks will continue monitoring rather than terminating the account.
By contrast, a merchant combining warning-letter exposure with rising disputes, aggressive continuity billing, and fulfillment delays often triggers reserve escalation or shutdown because the total operational risk profile deteriorates simultaneously.
Underwriting decisions are rarely based on one variable alone.
Claims Drift: The Hidden Risk Most Merchants Miss

One of the most common supplement underwriting problems is claims drift.
A merchant may begin relatively compliant, then gradually become more aggressive over time through:
- affiliate marketing
- ad agencies
- email campaigns
- social media creatives
- SEO content expansion
- influencer partnerships
Eventually the processor re-scans the merchant ecosystem and discovers language that materially changes the risk profile.
Processors frequently discover claims drift during periodic website rescans, affiliate audits, or chargeback investigations months after the original underwriting approval.
This is one reason many supplement merchant accounts are terminated months after approval.
The bank approved one risk profile.
The account evolved into another.
Can Merchants Still Get Approved After an FDA Warning Letter?
Yes — in many cases supplement merchants can still secure stable payment processing after an FDA warning letter.
But approval quality depends heavily on remediation.
Underwriters typically evaluate:
- whether claims were corrected
- how quickly remediation occurred
- chargeback stability
- current website compliance
- billing transparency
- fulfillment consistency
- management responsiveness
- whether the merchant disclosed the issue proactively
Underwriting note: Merchants that respond quickly, clean up claims exposure, stabilize fulfillment, and maintain dispute ratios below monitoring thresholds are often viewed more favorably than merchants who attempt to minimize or hide obvious compliance issues during underwriting review.
Strong underwriting positioning can materially improve outcomes.
This is one reason supplement businesses frequently work with underwriting-aware brokers instead of applying blindly through mainstream aggregators.
Common Mistakes Merchants Make After a Warning Letter
Applying Everywhere Immediately
Mass applying to multiple processors after a compliance issue often worsens placement difficulty.
Repeated declines create additional underwriting friction.
Leaving Aggressive Marketing Live
Many merchants remove claims from product pages while forgetting:
- old blogs
- affiliate pages
- email flows
- paid ads
- social media posts
Processors often scan far beyond the homepage.
Ignoring Chargeback Trends
Chargebacks frequently transform a compliance issue into a processor problem.
Processors tolerate regulatory complexity more comfortably when dispute ratios remain stable.
Using Mainstream Aggregators
Platforms like Stripe and Square rely heavily on automated enforcement systems and restricted-business monitoring policies.4
That structure rarely works well for nuanced high-risk supplement underwriting.
How to Reduce Payment Risk After an FDA Warning Letter
1. Tighten Claims Language
Review:
- product pages
- funnels
- affiliate content
- email campaigns
- social advertising
Claims exposure extends beyond the main website.
2. Stabilize Chargebacks
Dispute ratios strongly influence processor decisions after compliance events.
Monitor:
- refund trends
- descriptor confusion
- shipping delays
- subscription complaints
3. Improve Fulfillment Consistency
Shipping problems create underwriting problems.
Delayed fulfillment often leads directly to non-receipt disputes and elevated monitoring.
4. Maintain Documentation
Keep:
- COAs
- compliance updates
- policy revisions
- customer support logs
- processor communications
5. Use Underwriting-Aware Placement
Most supplement merchant account failures are compatibility failures.
The right acquiring bank matters more than the fastest approval.
Businesses seeking stable supplement merchant accounts should also review:
- nutraceutical payment processing compliance
- high-risk merchant accounts
- supplement merchant account shutdown risk
Where VERIFIED Fits
VERIFIED operates as a multi-bank high-risk broker rather than a single processor.
Different acquiring banks tolerate different supplement risk profiles. Some avoid weight-loss supplements entirely. Others allow them with enhanced underwriting controls, reserves, or monitoring requirements.
VERIFIED helps supplement merchants:
- position applications appropriately
- reduce underwriting friction
- structure backup processing relationships
- align gateways with risk profiles
- improve long-term account durability
In supplements, long-term stability usually depends more on underwriting compatibility than approval speed.
Further Reading
- Visa Chargeback Management Guidelines
- Visa Monitoring & Risk Programs
- FDA Warning Letters Database
- Stripe Restricted Businesses Policy
- FTC Health Claims Guidance
If your supplement business has experienced reserve increases, enhanced monitoring, or underwriting pressure following compliance concerns, addressing those issues before reapplying materially improves long-term processing stability.
Working with an underwriting-aware high-risk broker is often the difference between temporary approval and durable processing infrastructure.
Frequently Asked Questions
Can you still get a supplement merchant account after an FDA warning letter?
Yes. Many supplement merchants can still secure stable payment processing after an FDA warning letter if claims exposure is remediated properly, chargebacks remain controlled, and underwriting is handled strategically.
Why do payment processors care about FDA warning letters?
Processors view FDA warning letters as underwriting risk signals that may predict future disputes, reputational exposure, compliance costs, or chargeback increases.
Will an FDA warning letter automatically shut down my merchant account?
Not always. Some merchants continue processing successfully after remediation, while others face reserves, enhanced monitoring, or termination depending on the acquiring bank and overall risk profile.
What supplement claims create the most processor concern?
Weight-loss claims, disease-treatment language, before-and-after marketing, aggressive continuity billing funnels, and misleading medical positioning create the highest underwriting concern for supplement merchants.
What is claims drift in supplement payment processing?
Claims drift occurs when marketing language gradually becomes more aggressive over time through affiliates, agencies, blogs, or advertising campaigns, increasing underwriting and compliance risk.
Why are supplement merchants considered high-risk by processors?
Supplement merchants are considered high-risk because acquiring banks evaluate potential dispute exposure, compliance risk, continuity billing issues, and reputational concerns associated with aggressive marketing or elevated chargeback ratios.
Continue Reading
Let us discover your best options.
Either submit the form below or get in touch with an agent now (415) 835-4135.
Applying is risk-free; we send your details to underwriters to find the best fit and contact you with the best option or request more details, with no credit checks or commitments.












